Abstract
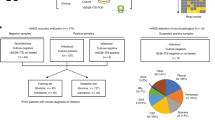
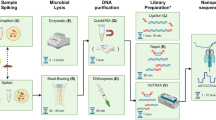
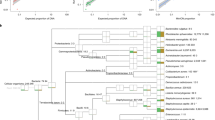
The gold standard for clinical diagnosis of bacterial lower respiratory infections (LRIs) is culture, which has poor sensitivity and is too slow to guide early, targeted antimicrobial therapy. Metagenomic sequencing could identify LRI pathogens much faster than culture, but methods are needed to remove the large amount of human DNA present in these samples for this approach to be feasible. We developed a metagenomics method for bacterial LRI diagnosis that features efficient saponin-based host DNA depletion and nanopore sequencing. Our pilot method was tested on 40 samples, then optimized and tested on a further 41 samples. Our optimized method (6 h from sample to result) was 96.6% sensitive and 41.7% specific for pathogen detection compared with culture and we could accurately detect antibiotic resistance genes. After confirmatory quantitative PCR and pathobiont-specific gene analyses, specificity and sensitivity increased to 100%. Nanopore metagenomics can rapidly and accurately characterize bacterial LRIs and might contribute to a reduction in broad-spectrum antibiotic use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All clinical sample sequence data and assemblies are available via European Nucleotide Archive (ENA) under study accession number PRJEB30781.
References
National Institute for Health and Care Excellence (NICE). Respiratory Tract Infections (Self-limiting): Prescribing Antibiotics NICE Clinical Guideline 69 (Centre for Clinical Practice, 2008); https://www.nice.org.uk/guidance/cg69
Chalmers, J. et al. Community-acquired pneumonia in the United Kingdom: a call to action. Pneumonia 9, 15 (2017).
Enne, V. I., Personne, Y., Grgic, L., Gant, V. & Zumla, A. Aetiology of hospital-acquired pneumonia and trends in antimicrobial resistance. Curr. Opin. Pulm. Med. 20, 252–258 (2014).
Carroll, K. C. Laboratory diagnosis of lower respiratory tract infections: controversy and conundrums. J. Clin. Microbiol. 40, 3115–3120 (2002).
Kollef, M. H. Microbiological diagnosis of ventilator-associated pneumonia. Am. J. Respir. Crit. Care Med. 173, 1182–1184 (2006).
Moran, G. J., Rothman, R. E. & Volturo, G. A. Emergency management of community-acquired bacterial pneumonia: what is new since the 2007 Infectious Diseases Society of America/American Thoracic Society guidelines. Am. J. Emerg. Med. 31, 602–612 (2013).
Garcin, F. et al. Non-adherence to guidelines: an avoidable cause of failure of empirical antimicrobial therapy in the presence of difficult-to-treat bacteria. Intensive Care Med. 36, 75–82 (2010).
Lim, W. S. et al. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax 64, iii1 (2009).
Burnham, C. A. & Carroll, K. C. Diagnosis of Clostridium difficile infection: an ongoing conundrum for clinicians and for clinical laboratories. Clin. Microbiol. Rev. 26, 604–630 (2013).
Lees, E. A., Miyajima, F., Pirmohamed, M. & Carrol, E. D. The role of Clostridium difficile in the paediatric and neonatal gut—a narrative review. Eur. J. Clin. Microbiol. Infect. Dis. 35, 1047–1057 (2016).
Cookson, W. O. C. M., Cox, M. J. & Moffatt, M. F. New opportunities for managing acute and chronic lung infections. Nat. Rev. Microbiol. 16, 111 (2017).
Davies, S. C. in Annual Report of the Chief Medical Officer Ch. 1 (Department of Health, 2017); https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/631043/CMO_annual_report_generation_genome.pdf
UK Goverment. Tackling antimicrobial resistance 2019–2024. The UK’s five-year national action plan. (2019).
O’Neill, J. Tackling drug-resistant infections globally: final report and recommendations. in The Review on Microbial Resistance 1–84 (2016).
Fukumoto, H., Sato, Y., Hasegawa, H., Saeki, H. & Katano, H. Development of a new real-time PCR system for simultaneous detection of bacteria and fungi in pathological samples. Int. J. Clin. Exp. Pathol. 8, 15479–15488 (2015).
Hassibi, A. et al. Multiplexed identification, quantification and genotyping of infectious agents using a semiconductor biochip. Nat. Biotechnol. 36, 738–745 (2018).
Kais, M., Spindler, C., Kalin, M., Örtqvist, Å. & Giske, C. G. Quantitative detection of Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis in lower respiratory tract samples by real-time PCR. Diagn. Microbiol. Infect. Dis. 55, 169–178 (2006).
Kodani, M. et al. Application of TaqMan low-density arrays for simultaneous detection of multiple respiratory pathogens. J. Clin. Microbiol. 49, 2175–2182 (2011).
Hayon, J. A. N. et al. Role of serial routine microbiologic culture results in the initial management of ventilator-associated pneumonia. Am. J. Respir. Crit. Care Med. 165, 41–46 (2002).
Buchan, B. W. & Ledeboer, N. A. Emerging technologies for the clinical microbiology laboratory. Clin. Microbiol. Rev. 27, 783 (2014).
Huang, T.-D. et al. Analytical validation of a novel high multiplexing real-time PCR array for the identification of key pathogens causative of bacterial ventilator-associated pneumonia and their associated resistance genes. J. Antimicrob. Chemother. 68, 340–347 (2012).
Chiu, C. Y. & Miller, S. A. Clinical metagenomics. Nat. Rev. Genet. 20, 341–355 (2019).
Loman, N. J. et al. Performance comparison of benchtop high-throughput sequencing platforms. Nat. Biotechnol. 30, 434 (2012).
Strauch, B. et al. LiveKraken––real-time metagenomic classification of illumina data. Bioinformatics 34, 3750–3752 (2018).
Faria, N. R. et al. Establishment and cryptic transmission of Zika virus in Brazil and the Americas. Nature 546, 406 (2017).
Quick, J. et al. Real-time, portable genome sequencing for Ebola surveillance. Nature 530, 228 (2016).
Greninger, A. L. et al. Rapid metagenomic identification of viral pathogens in clinical samples by real-time nanopore sequencing analysis. Genome Med. 7, 99 (2015).
Schmidt, K. et al. Identification of bacterial pathogens and antimicrobial resistance directly from clinical urines by nanopore-based metagenomic sequencing. J. Antimicrob. Chemother. 72, 104–114 (2017).
Pendleton, K. M. et al. Rapid pathogen identification in bacterial pneumonia using real-time metagenomics. Am. J. Respir. Crit. Care Med. 196, 1610–1612 (2017).
Feehery, G. R. et al. A method for selectively enriching microbial DNA from contaminating vertebrate host DNA. PLoS ONE 8, e76096 (2013).
Hasan, M. R. et al. Depletion of human DNA in spiked clinical specimens for improvement of sensitivity of pathogen detection by next-generation sequencing. J. Clin. Microbiol. 54, 919–927 (2016).
Marotz, C. A. et al. Improving saliva shotgun metagenomics by chemical host DNA depletion. Microbiome 6, 42 (2018).
Zelenin, S. et al. Microfluidic-based isolation of bacteria from whole blood for sepsis diagnostics. Biotechnol. Lett. 37, 825–830 (2015).
Couto, N. et al. Critical steps in clinical shotgun metagenomics for the concomitant detection and typing of microbial pathogens. Sci. Rep. 8, 13767 (2018).
McIntosh, J. Emergency pathology service. Lancet 247, 669–670 (1946).
Martner, A., Dahlgren, C., Paton, J. C. & Wold, A. E. Pneumolysin released during Streptococcus pneumoniae autolysis is a potent activator of intracellular oxygen radical production in neutrophils. Infect. Immun. 76, 4079–4087 (2008).
Chen, J. H. K. et al. Use of MALDI biotyper plus ClinProTools mass spectra analysis for correct identification of Streptococcus pneumoniae and Streptococcus mitis. J. Clin. Pathol. 68, 652–656 (2015).
Kutlu, S. S., Sacar, S., Cevahir, N. & Turgut, H. Community-acquired Streptococcus mitis meningitis: a case report. Int. J. Infect. Dis. 12, e107–e109 (2008).
Langelier, C. et al. Integrating host response and unbiased microbe detection for lower respiratory tract infection diagnosis in critically ill adults. Proc. Natl Acad. Sci. USA 115, E12353 (2018).
Eliopoulos, G. M. & Huovinen, P. Resistance to trimethoprim-sulfamethoxazole. Clin. Infect. Dis. 32, 1608–1614 (2001).
Enne, V. I., King, A., Livermore, D. M. & Hall, L. M. C. Sulfonamide resistance in Haemophilus influenzae mediated by acquisition of sul2 or a short insertion in chromosomal folP. Antimicrob. Agents Chemother. 46, 1934–1939 (2002).
Ashton, P. M. et al. MinION nanopore sequencing identifies the position and structure of a bacterial antibiotic resistance island. Nat. Biotechnol. 33, 296 (2014).
Orlek, A. et al. Plasmid classification in an era of whole-genome sequencing: application in studies of antibiotic resistance epidemiology. Front. Microbiol. 8, 182 (2017).
Xia, Y. et al. MinION nanopore sequencing enables correlation between resistome phenotype and genotype of coliform bacteria in municipal sewage. Front. Microbiol. 8, 2105 (2017).
Leggett, R. M. et al. Rapid MinION metagenomic profiling of the preterm infant gut microbiota to aid in pathogen diagnostics. Preprint at biorxiv https://www.biorxiv.org/content/10.1101/180406v1 (2017).
Roberts, A. P. & Mullany, P. Tn916-like genetic elements: a diverse group of modular mobile elements conferring antibiotic resistance. FEMS Microbiol. Rev. 35, 856–871 (2011).
Santoro, F., Vianna, M. E. & Roberts, A. P. Variation on a theme; an overview of the Tn916/Tn1545 family of mobile genetic elements in the oral and nasopharyngeal streptococci. Front. Microbiol. 5, 535 (2014).
Tantivitayakul, P., Lapirattanakul, J., Vichayanrat, T. & Muadchiengka, T. Antibiotic resistance patterns and related mobile genetic elements of pneumococci and β-hemolytic streptococci in Thai healthy children. Indian J. Microbiol. 56, 417–425 (2016).
Deurenberg, R. H. et al. Application of next generation sequencing in clinical microbiology and infection prevention. J. Biotechnol. 243, 16–24 (2017).
Greninger, A. L. et al. Rapid metagenomic next-generation sequencing during an investigation of hospital-acquired human parainfluenza virus 3 infections. J. Clin. Microbiol. 55, 177–182 (2017).
Services Unit, Microbiology Services, Public Health England. UK standards for microbiology investigations: investigation of bronchoalveolar lavage, sputum and associated specimens. Bacteriology B57, 38 (2018).
Anscombe, C., Misra, R. V. & Gharbia, S. Whole genome amplification and sequencing of low cell numbers directly from a bacteria spiked blood model. Preprint at biorxiv https://www.biorxiv.org/content/10.1101/153965v3(2018).
Kim, D., Song, L., Breitwieser, F. P. & Salzberg, S. L. Centrifuge: rapid and sensitive classification of metagenomic sequences. Genome Res. 26, 1721–1729 (2016).
Jia, B. et al. CARD 2017: expansion and model-centric curation of the comprehensive antibiotic resistance database. Nucleic Acids Res. 45, D566–D573 (2017).
Leggett, R. M., Heavens, D., Caccamo, M., Clark, M. D. & Davey, R. P. NanoOK: multi-reference alignment analysis of nanopore sequencing data, quality and error profiles. Bioinformatics 32, 142–144 (2015).
Price, E. P. et al. Simultaneous identification of Haemophilus influenzae and Haemophilus haemolyticus using real-time PCR. Future Microbiol. 12, 585–593 (2017).
Li, H. Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics 34, 3094–3100 (2018).
Koren, S., Walenz, B. P., Berlin, K., Miller, J. R. & Phillippy, A. M. Canu: scalable and accurate long-read assembly via adaptive k-mer weighting and repeat separation. Genome Res. 27, 722–736 (2017).
Koren, S. et al. De novo assembly of haplotype-resolved genomes with trio binning. Nat. Biotechnol. 36, 1174–1182 (2018).
Alikhan, N.-F., Petty, N. K., Ben Zakour, N. L. & Beatson, S. A. BLAST ring image generator (BRIG): simple prokaryote genome comparisons. BMC Genomics 12, 402 (2011).
Acknowledgements
This paper presents independent research funded by the National Institute for Health Research (NIHR) under its Program Grants for Applied Research Program (reference no. RP-PG-0514-20018, J.O.G., D.M.L., R.B. and H.R.), the UK Antimicrobial Resistance Cross Council Initiative (no. MR/N013956/1, J.O.G. and G.L.K.), Rosetrees Trust (no. A749, J.O.G.), the University of East Anglia (to J.O.G. and T.C.), Oxford Nanopore Technologies (to J.O.G., T.C., A.A. and D.J.T.), the Biotechnology and Biological Sciences Research Council (BBSRC) Institute Strategic Programme Microbes in the Food Chain BB/R012504/1 and its constituent projects BBS/E/F/000PR10348 and BBS/E/F/000PR10349 (J.O.G., J.W. and G.L.K.), MRC Doctoral Antimicrobial Research Training (DART) Industrial CASE Programme grant number MR/R015937/1 (J.O.G. and A.A.) and BBSRC grants (nos. BB/N023196/1 and BB/CSP17270/1, to R.M.L.). Part of the bioinformatics analysis was run on CLIMB-computing servers, an infrastructure supported by a grant from the UK Medical Research Council (no. MR/L015080/1).
Author information
Authors and Affiliations
Contributions
The study was devised by J.O.G., J.W. and D.J.T. Laboratory work and data analysis were performed by T.C., G.L.K., A.A., H.R., R.B., D.M.L., R.M.L. and J.O.G. Clinical samples were collected and analyzed by C.J., S.G. and D.R. All authors contributed to writing and reviewing the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.O.G., R.M.L., G.L.K. and T.C. received financial support for attending ONT and other conferences and/or an honorarium for speaking at ONT headquarters. J.O.G., A.A. and T.C. received funding and consumable support from ONT for PhD studentships. D.J.T. is a full-time employee and share-option holder of Oxford Nanopore Technologies Ltd. R.M.L. and J.O.G. received free flow cells as part of the MAP and MARC programs.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Fig. 1, Supplementary Tables 1–9
Rights and permissions
About this article
Cite this article
Charalampous, T., Kay, G.L., Richardson, H. et al. Nanopore metagenomics enables rapid clinical diagnosis of bacterial lower respiratory infection. Nat Biotechnol 37, 783–792 (2019). https://doi.org/10.1038/s41587-019-0156-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-019-0156-5
This article is cited by
-
Diagnostic performance of metagenomic next-generation sequencing for the detection of pathogens in cerebrospinal fluid in pediatric patients with central nervous system infection: a systematic review and meta-analysis
BMC Infectious Diseases (2024)
-
Antimicrobial resistance prediction by clinical metagenomics in pediatric severe pneumonia patients
Annals of Clinical Microbiology and Antimicrobials (2024)
-
Development and proof-of-concept demonstration of a clinical metagenomics method for the rapid detection of bloodstream infection
BMC Medical Genomics (2024)
-
mEnrich-seq: methylation-guided enrichment sequencing of bacterial taxa of interest from microbiome
Nature Methods (2024)
-
Short turnaround time of seven to nine hours from sample collection until informed decision for sepsis treatment using nanopore sequencing
Scientific Reports (2024)