Future Perspective: From Optoelectronics to Nanomedicine
The last decade has witnessed the development of QD–CNT complexes for several applications from optoelectronics to biosensing (Table 3). From electronics[101] to biology,[102] the potential applications of QD–CNT complexes are immense.
Currently, most experiments on the conjugation of QDs to CNTs are in the realm optoelectronics,[103–105] including field emission displays,[106] computer memory devices,[107] photovoltaic cells[108,109] and photocatalytic devices.[110,111] The combination of these two seemingly disparate nanomaterials has indeed allowed improved electronic and optical properties, because the electronic transport effects can be tuned from weak to strong by simply changing gate voltages,[112] while optical signals are greatly enhanced by highly photostable QDs exhibiting quantum yields close to unity.
Mounting evidence suggests that the conjugation of QDs to CNTs allow them to function as novel hybrid nanocomposite materials,[113–115] displaying unique physical[116–118] and biological[119–122] properties (Figure 7). These QD–CNT complexes can be used in intracellular fluorescent imaging[123–126] as they have electrochemiluminescent (ECL) abilities.[127] Furthermore, QD–CNT complexes can also function as biosensors,[128,129] biological nanoprobes,[130–132] as well as loading and delivery of nanoparticles into cells.[133,134]
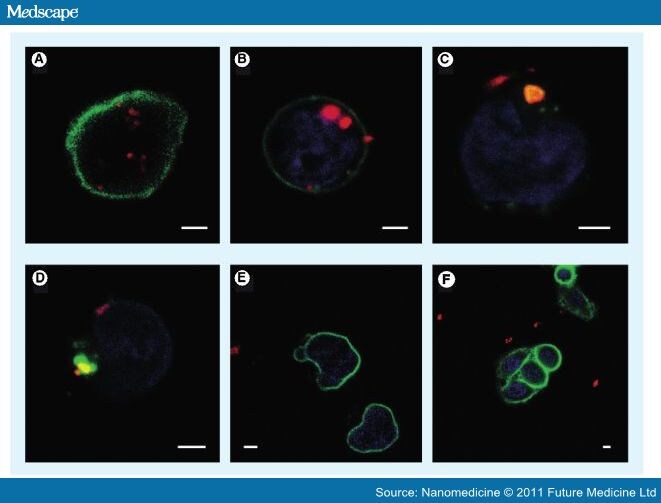
Figure 7.
Fluorescent images of quantum dot–streptavidin–carbon nanotubes complex taken up into cancer cells. (A) Confocal image of QD–Str–CNT (red) taken up by Jurkat T leukemia cells via biotinylated aCD3-CD3 receptor-mediated endocytosis. (B) Confocal image with DAPI to visualize nuclear region (blue). (C & D) Confocal images visualizing QD–Str–CNT complex (red), nuclear region (blue) and lysosome (green). (E & F) No uptake of QD–Str–CNT complex when cells were incubated with unconjugated (no biotin) aCD3, indicating a CD3 receptor-mediated endocytosis. Note: Str binds strongly to biotin. (A–F) Scale bars represent 2 µm.
CNT: Carbon nanotube; QD: Quantum dot; Str: Streptavidin.
Reproduced with permission from [119]. ACS Publications © 2006.
It has been suggested that different QDs can be conjugated to a drug–CNT molecule, and it can be designed to fluoresce at different colors to indicate different biological activities[135] (e.g., position, delivery and cellular uptake of the drug). Taken together, there is indeed strong evidence that novel properties can be derived from the conjugation of QDs to CNTs, with potential application in cancer theranostics.
Despite such undeniable contribution, the combination of functionalized CNTs and QDs for other biomedical applications is severely under-represented; for example, drug delivery of small molecules (e.g., anticancer, antiviral, or antibacterial agents) from QD–CNT constructs is still vastly unexplored. This could be attributed to the fact that even with multiple functionalization, some surfaces of CNTs remain restricted, thus severely limiting the amount of drug that could potentially be loaded. Moreover, there seems to be a prevailing skepticism towards extensive use of these constructs in nanomedicine, mainly due to conflicting toxicological studies on pristine and uncoated CNTs, which poses the question of its biocompatibility.
Nevertheless, once efficiently combined, these nanomaterials display fascinating properties that envisage potential benefits to cancer medicine, both in terms of therapy and diagnosis. CNTs and QDs represent worthwhile research tools in cancer treatment because they both exhibit high mechanical stability and nanometric dimensions, which enable penetration into normally inaccessible tumor sites, while minimizing the probability of being engulfed by macrophages (i.e., a 'stealth' effect). In addition, escape from the normal phagocytic defenses could be associated with a sustained presence in blood circulation (especially in the form of PEGylated systems), with concomitant prolonged effects of enhanced cellular uptake. Indeed, CNTs have shown the ability to be easily internalized by cells and accumulate at the tumor site due to the enhanced permeability retention effect.[77]
In contrast to spherical nanoparticles, the long and cylindrical shape of CNTs present a large inner volume that could be filled with a plethora of biomolecules ranging from small derivatives to proteins.[136] This introduces the possibility of drug loading into the internal cylindrical area, while conferring chemical properties via their external wall functionalization with QDs and/or solubilizing agents, thus rendering the constructs in a biocompatible form.
Strategic targeting moieties (e.g., folic acid (FA) or HER2) onto CNTs' surfaces could increase the specificity of targeting cancer cells.[137] For instance, tumor cells generally show increased FA receptors on their surfaces, and this can be exploited by functionalizing CNTs with FA to confer increased specificity in killing cancer cells.
Biological systems are highly transparent to 700–1100 nm NIR light. CNTs have been demonstrated to intrinsically display strong optical absorbance in this spectral window, and this could be used to trigger endosomal rupture of cancer cells via NIR laser pulses. Alternatively, radiofrequency waves can intercalate deeply in the body whilst minimizing damage to the surrounding tissue.[96] Thus, if injected correspondingly to a specific tumor, these nanosystems can selectively eliminate cancerous cells.
Overall, the key aspects in the conjugation of QDs to CNTs for future biomedical applications are the development of nanosystems presenting: one or more therapeutic molecules with recognition capacity; optical signals for imaging; and specific targeting groups within the same multifaceted entity. On the basis of the data gathered thus far with regard to nanoparticles, QD–CNT constructs in particular seem to embody the hallmarks of a promising biomedical research tool, with particular relevance in cancer theranostics.
Nanomedicine. 2011;6(6):1101-1114. © 2011 Future Medicine Ltd.
Cite this: Quantum Dots and Carbon Nanotubes in Oncology - Medscape - Aug 01, 2011.




Comments