Abstract
Exposure of lung tissues to cigarette smoke is a major cause of human disease and death worldwide. Unfortunately, adequate model systems that can reliably recapitulate disease biogenesis in vitro, including exposure of the human lung airway to fresh whole cigarette smoke (WCS) under physiological breathing airflow, are lacking. This protocol extension builds upon, and can be used with, our earlier protocol for microfabrication of human organs-on-chips. Here, we describe the engineering, assembly and operation of a microfluidically coupled, multi-compartment platform that bidirectionally ‘breathes’ WCS through microchannels of a human lung small airway microfluidic culture device, mimicking how lung cells may experience smoke in vivo. Several WCS-exposure systems have been developed, but they introduce smoke directly from above the cell cultures, rather than tangentially as naturally occurs in the lung due to lateral airflow. We detail the development of an organ chip–compatible microrespirator and a smoke machine to simulate breathing behavior and smoking topography parameters such as puff time, inter-puff interval and puffs per cigarette. Detailed design files, assembly instructions and control software are provided. This novel platform can be fabricated and assembled in days and can be used repeatedly. Moderate to advanced engineering and programming skills are required to successfully implement this protocol. When coupled with the small airway chip, this protocol can enable prediction of patient-specific biological responses in a matched-comparative manner. We also demonstrate how to adapt the protocol to expose living ciliated airway epithelial cells to smoke generated by electronic cigarettes (e-cigarettes) on-chip.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Powell, H. A., Iyen-Omofoman, B., Baldwin, D. R., Hubbard, R. B. & Tata, L. J. Chronic obstructive pulmonary disease and risk of lung cancer: the importance of smoking and timing of diagnosis. J. Thorac. Oncol. 8, e34 (2013).
Benam, K. H. et al. Matched-comparative modeling of normal and diseased human airway responses using a microengineered breathing lung chip. Cell Syst. 3, 456–466.e454 (2016).
Bhatia, S. N. & Ingber, D. E. Microfluidic organs-on-chips. Nat. Biotechnol. 32, 760–772 (2014).
Benam, K. H. et al. Small airway-on-a-chip enables analysis of human lung inflammation and drug responses in vitro. Nat. Methods 13, 151–157 (2016).
Benam, K. H. & Ingber, D. E. Commendation for exposing key advantage of organ chip approach. Cell Syst. 3, 411 (2016).
Benam, K. H., Konigshoff, M. & Eickelberg, O. Breaking the in vitro barrier in respiratory medicine. engineered microphysiological systems for chronic obstructive pulmonary disease and beyond. Am. J. Respir. Crit. Care Med. 197, 869–875 (2018).
Niemeyer, B. F., Zhao, P., Tuder, R. M. & Benam, K. H. Advanced microengineered lung models for translational drug discovery. SLAS Discov 23, 777–789 (2018).
Benam, K. H. Disrupting experimental strategies for inhalation toxicology: the emergence of microengineered breathing-smoking human lung-on-a-chip. Appl. In Vitro Toxicol. 4, 107–114 (2018).
Benam, K. H. et al. Exploring new technologies in biomedical research. Drug Discov. Today 24, 1242–1247 (2019).
Novak, R. et al. Scalable fabrication of stretchable, dual channel, microfluidic organ chips. J. Vis. Exp. 140, e58151 (2018).
Huh, D. et al. Microfabrication of human organs-on-chips. Nat. Protoc. 8, 2135–2157 (2013).
Benam, K. H. et al. Human lung small airway-on-a-chip protocol. Methods Mol. Biol. 1612, 345–365 (2017).
Gotts, J. E. et al. Cigarette smoke exposure worsens endotoxin-induced lung injury and pulmonary edema in mice. Nicotine Tob. Res. 19, 1033–1039 (2017).
Sundar, I. K., Nevid, M. Z., Friedman, A. E. & Rahman, I. Cigarette smoke induces distinct histone modifications in lung cells: implications for the pathogenesis of COPD and lung cancer. J. Proteome Res. 13, 982–996 (2014).
Thorne, D. & Adamson, J. A review of in vitro cigarette smoke exposure systems. Exp. Toxicol. Pathol. 65, 1183–1193 (2013).
Thorne, D. et al. The mutagenic assessment of mainstream cigarette smoke using the Ames assay: a multi-strain approach. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 782, 9–17 (2015).
Thorne, D. et al. Characterisation of a Vitrocell(R) VC 10 in vitro smoke exposure system using dose tools and biological analysis. Chem. Cent. J. 7, 146 (2013).
Thorne, D., Larard, S., Baxter, A., Meredith, C. & Gaa, M. The comparative in vitro assessment of e-cigarette and cigarette smoke aerosols using the gammaH2AX assay and applied dose measurements. Toxicol. Lett. 265, 170–178 (2017).
Mathis, C. et al. Human bronchial epithelial cells exposed in vitro to cigarette smoke at the air–liquid interface resemble bronchial epithelium from human smokers. Am. J. Physiol. Lung Cell. Mol. Physiol. 304, L489–L503 (2013).
Phillips, J., Kluss, B., Richter, A. & Massey, E. Exposure of bronchial epithelial cells to whole cigarette smoke: assessment of cellular responses. Altern. Lab. Anim. 33, 239–248 (2005).
Scian, M. J., Oldham, M. J., Kane, D. B., Edmiston, J. S. & McKinney, W. J. Characterization of a whole smoke in vitro exposure system (Burghart Mimic Smoker-01). Inhal. Toxicol. 21, 234–243 (2009).
Glader, P. et al. Cigarette smoke extract modulates respiratory defence mechanisms through effects on T-cells and airway epithelial cells. Respir. Med. 100, 818–827 (2006).
Mio, T. et al. Cigarette smoke induces interleukin-8 release from human bronchial epithelial cells. Am. J. Respir. Crit. Care Med. 155, 1770–1776 (1997).
Hellermann, G. R., Nagy, S. B., Kong, X., Lockey, R. F. & Mohapatra, S. S. Mechanism of cigarette smoke condensate-induced acute inflammatory response in human bronchial epithelial cells. Respir. Res. 3, 22 (2002).
Anto, R. J., Mukhopadhyay, A., Shishodia, S., Gairola, C. G. & Aggarwal, B. B. Cigarette smoke condensate activates nuclear transcription factor-kappaB through phosphorylation and degradation of IkappaB(alpha): correlation with induction of cyclooxygenase-2. Carcinogenesis 23, 1511–1518 (2002).
Carnevali, S. et al. Cigarette smoke extract induces oxidative stress and apoptosis in human lung fibroblasts. Am. J. Physiol. Lung Cell. Mol. Physiol. 284, L955–L963 (2003).
Ween, M. P., Whittall, J. J., Hamon, R., Reynolds, P. N. & Hodge, S. J. Phagocytosis and Inflammation: exploring the effects of the components of E-cigarette vapor on macrophages. Physiol. Rep. 5, e13370 (2017).
Hoyt, J. C. et al. Cigarette smoke decreases inducible nitric oxide synthase in lung epithelial cells. Exp. Lung Res. 29, 17–28 (2003).
Adamson, J., Haswell, L. E., Phillips, G., & Gaça, M. D. In vitro models of chronic obstructive pulmonary disease (COPD). In Bronchitis (ed. Martín-Loeches, I.) (InTechOpen, 2011).
Rao, P., Ande, A., Sinha, N., Kumar, A. & Kumar, S. Effects of cigarette smoke condensate on oxidative stress, apoptotic cell death, and HIV replication in human monocytic cells. PLoS ONE 11, (e0155791 (2016).
Strasser, A. A. et al. Nicotine metabolite ratio predicts smoking topography and carcinogen biomarker level. Cancer Epidemiol. Biomarkers Prev. 20, 234–238 (2011).
Hammond, D., Fong, G. T., Cummings, K. M. & Hyland, A. Smoking topography, brand switching, and nicotine delivery: results from an in vivo study. Cancer Epidemiol. Biomarkers Prev. 14, 1370–1375 (2005).
Manigrasso, M., Buonanno, G., Fuoco, F. C., Stabile, L. & Avino, P. Aerosol deposition doses in the human respiratory tree of electronic cigarette smokers. Environ. Pollut. 196, 257–267 (2015).
Benam, K. H., Kok, W. L., McMichael, A. J. & Ho, L. P. Alternative spliced CD1d transcripts in human bronchial epithelial cells. PLoS ONE 6, e22726 (2011).
Huang da, W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4, 44–57 (2009).
Harvey, B. G. et al. Modification of gene expression of the small airway epithelium in response to cigarette smoking. J. Mol. Med. 85, 39–53 (2007).
Keatings, V. M., Collins, P. D., Scott, D. M. & Barnes, P. J. Differences in interleukin-8 and tumor necrosis factor-alpha in induced sputum from patients with chronic obstructive pulmonary disease or asthma. Am. J. Respir. Crit. Care Med. 153, 530–534 (1996).
Dickens, J. A. et al. COPD association and repeatability of blood biomarkers in the ECLIPSE cohort. Respir. Res. 12, 146 (2011).
Imai, K. et al. Human collagenase (matrix metalloproteinase-1) expression in the lungs of patients with emphysema. Am. J. Respir. Crit. Care Med. 163, 786–791 (2001).
Mercer, B. A., Kolesnikova, N., Sonett, J. & D’Armiento, J. Extracellular regulated kinase/mitogen activated protein kinase is up-regulated in pulmonary emphysema and mediates matrix metalloproteinase-1 induction by cigarette smoke. J. Biol. Chem. 279, 17690–17696 (2004).
Stanley, P. J., Wilson, R., Greenstone, M. A., MacWilliam, L. & Cole, P. J. Effect of cigarette smoking on nasal mucociliary clearance and ciliary beat frequency. Thorax 41, 519–523 (1986).
Yaghi, A., Zaman, A., Cox, G. & Dolovich, M. B. Ciliary beating is depressed in nasal cilia from chronic obstructive pulmonary disease subjects. Respir. Med. 106, 1139–1147 (2012).
Zhou, H. et al. Increased nasal epithelial ciliary beat frequency associated with lifestyle tobacco smoke exposure. Inhal. Toxicol. 21, 875–881 (2009).
Acknowledgements
This research was sponsored by the Wyss Institute for Biologically Inspired Engineering at Harvard University and the Defense Advanced Research Projects Agency under Cooperative Agreement no. W911NF-12-2-0036. The views and conclusions contained in this document are those of the authors and should not be interpreted as representing the official policies, either expressed or implied, of the Defense Advanced Research Projects Agency or the U.S. Government.
Author information
Authors and Affiliations
Contributions
K.H.B. and R.N. led system and protocol development and conducted smoking studies; R.N. and Y.C. designed the smoking robot and control software; T.C.F. and R.N. designed the microrespirator; K.H.B., R.N. and D.E.I. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
D.E.I. is a founder and holds equity in Emulate, Inc. and chairs its scientific advisory board. K.H.B., R.N., Y.C. and D.E.I. are also inventors on intellectual property licensed to Emulate, Inc.
Additional information
Peer review information Nature Protocols thanks Amy Ryan (Firth) and other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key reference using this protocol
Benam, K. H. et al. Cell Syst. 3, 456–466.e4 (2016): https://doi.org/10.1016/j.cels.2016.10.003
This protocol is an extension to: Nat. Protoc. doi:10.1038/nprot.2013.137
Integrated supplementary information
Supplementary Fig. 2 Assembly of smoke exhaust and smoke containment modules.
a. The Smoke Exhaust Module is assembled by: i) mounting 1”x1” fans to mounts using m2 screws, ii) inserting bulkhead mount barbs into barb mounts, iii) acrylic solvent bonding the remaining acrylic walls (back, top, outer_1, outer_2, and inner). b. The Smoke Containment Module Doors (2) are assembled by acrylic solvent bonding: i) door handles to each acrylic front door and back door, ii) rail_1 and rail_2 parts to each other, iii) rail assembly to side wall. c. The Smoke Containment Module is assembled by acrylic solvent bonding Sidewalls (2), Front wall, Back wall, and Top wall, making sure walls are facing the correct orientations as shown in figure. d. The Smoke Exhaust and Containment are assembled by acrylic solvent bonding exhaust module to top wall of the Smoke Containment Module, making sure exhaust module is facing the correct orientation as shown in figure.
Supplementary Fig. 3 Assembly of smoke wheel, ignition, electronics, and mouthpiece modules.
a. The Smoke Wheel Module is assembled by: i) acrylic solvent bonding discs 1-4 (use alignment pinhole feature on each disc to keep holes concentric), ii) inserting 2 silicone gaskets in each hole of disc assembly, iii) in order, assembling ¼”-28 nut, ¼” washer, acrylic wheel assembly, ¼” washer, and ¼”-28 acorn nut onto partially threaded ¼” rod. b. The Electronics Module is comprised of two layers. The top layer houses the majority of the electronics, which consist of: 2 solid state relays, 1 stepper motor driver, 1 diaphragm pump, 1 Arduino Uno, 4 ¼” mounted bearings, 2 ¼” tooth gears, 1 ¼” shaft coupler, 2 tubing pinch valves, 1 5 mL disposable reservoir with cap, 1 stepper motor, ¼” shaft, and M3 screws to mount components onto electronics and motor shelf. Underneath the top shelf is the power supply and mouthpiece module. The top layer electronics are mounted to a removable acrylic board which sits on a shelf which is a fixed part of the Base Frame of the Smoke Generator Assembly. The components related to the motor drive are mounted to a surface which is a fixed part of the Base Frame of the Smoke Generator Assembly. The Smoke Wheel Module is then fixed through the concentrically aligned mounted bearings and gears, which then connect it to the motor drive components. c. Wire the electronic components as shown in this wiring diagram. Specific communication ports on the Arduino should agree with the control software. d. The Mouthpiece Module is assembled by passing through two partially threaded rods through the outer holes of the mounting plate. Then fasten the barb into the threaded Teflon mouthpiece through the Acrylic Mounting Plate, with the Teflon piece facing the same side as the threads. The Mouthpiece Module is then fixed into the Smoke Generator assembly by passing the two rods through two pairs of holes aligned concentrically on two walls of the Base Frame Module. The Mouthpiece Module can then be fixed in place by threading the rods into the tapped holes in the wall of the Base Frame Module.
Supplementary Fig. 4 Assembly of ignition module.
a. Ignition Main Body is 3d-printed. b. Carriage, rail, and linear actuator are mounted onto main body. c. NiChrome wire is coiled onto ceramic rod, which is then mounted into machined Teflon cylinder, using two screws to hold its place. Shoulder screw is screwed through Ignition Holder into center of Teflon cylinder. Another shoulder screw with a compression spring connects the Ignition Holder to the Ignition Main Body. d. Shoulder screws are aligned through slotted holes on lever, connecting the carriage and linear actuator to the main body of the ignition module. e. Assembled ignition module is mounted onto the Base Frame Assembly via. M6 screws and wingnuts. The NiChrome wire should be able to touch against the tip of a cigarette when linear actuator is fully actuated.
Supplementary Fig. 5 Front and rear view of base frame and smoke generator assembly.
a. The Base Frame is assembled by acrylic solvent bonding the acrylic Base Frame components together and screwing in support feet (4) and brackets (4) into base. b. The entire Smoke Generator is assembled by putting together all components from Figs. 2–5. The Smoke Containment Module can be locked onto the Base Frame by aligning screws and nuts through holes on the front and back walls of the Smoke Containment Module and brackets on Base Frame Module. The inside of the Smoke Generator can be accessed using the sliding doors from the Smoke Containment Module. Wires for the electronics and fed through an opening in the back wall of the Smoke Containment Module.
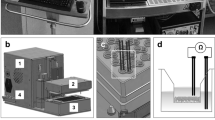
Supplementary Fig. 6 Assembly of microrespirator.
a. i) Attach corner brackets to aluminum extrusions uses t-slot nuts and bolts, ii) attach endMotor piece to corner brackets using nuts and bolts, iii) attach motors to endMotor piece using M3 screws, iv) attach shaft couplers to motor shaft by gently tightening set screw, v) attach corner brackets to aluminum extrusions as above, vi) attach endThrustBearing piece to corner rackets as above. b. i) Attach corner brackets to aluminum extrusions as above, ii) hold the syringeBase piece such that the holes for the syringes are vertically oriented, iii) place the syringes into these holes, such that they hang down by gravity, with the shoulder of the syringe resting on the syringeBase piece, iv) align the syringeBaseEtch piece and secure the assembly by inserting the central screws gently tightening their nuts. Secure this subassembly to corner brackets as above. c. i) Insert the GasketMaterialCircular pieces into the holes etched into the pistonEnd piece, ii) align the pistonHole piece with the pistonEnd-Hole piece and hold them together such that the circular holes oriented vertically, as above, and drop the pistons into the holes, iii) carefully align this subassembly with the pistonEnd piece containing the GasketMaterialCircular pieces, iv) Insert the thrust bearings and gently secure them with nuts and bolts. Attach the lead screw nuts gently, using nuts and bolts, v) the syringe pistons can now be aligned and inserted into the syringes. d. i) Insert the linear motion shafts through the syringeBase and syringeBaseEtch pieces, very loosely placing a collar clamps onto the shafts before they are inserted into the thrust bearings, through the endThrustBearing piece, and finally secured to the endMotor piece using screws, ii) terminal collar clamps can now be slid onto the ends and all collar clamps gently tightened, iii) threaded rods can be inserted, with a small collar clamp and thrust bearing put on each, before they pass through the pistonEnd piece. At this point, each threaded rod should have another thrust bearing and small collar clamp slid on before the threaded rod is secured by the shaft coupler, iv) the limit switch holder and limit switch can be attached, v) all pieces can be gently tightened, vi) the shelfSliders can be attached using t-slot nuts and bolts, vii) the fans should be oriented such that the air flow is directed at the motors and can be attached to the endMotor piece using zip ties. e. Wire the electronic components as shown in this wiring diagram. Specific communication ports on the Arduino should agree with the control software.
Supplementary Fig. 7 Inclusion of appropriate optimization and quality control steps when utilizing the biomimetic smoking robot.
a. Quantification of lactate dehydrogenase (LDH) release, as a marker of apparent cellular toxicity and death, when small airway chips were continuously (in the absence of inter-puff interval) were exposed to whole cigarette smoke from nine 3R4F cigarettes for 75 min (*p < 0.05). b. Levels of secreted matrix metallopeptidase 1 (MMP-1) in small airway chips lined with bronchiolar epithelial cells isolated from normal or COPD subjects in the presence (+) or absence (-) of breathing motions (Breathing) or exposure to whole cigarette smoke for 75 min (Smoking) (*p < 0.05, **p < 0.01, n.s.: not significant; data from one representative healthy subject and a representative COPD donor with 4–5 biological replicates per condition).
Supplementary information
Supplementary Information
Supplementary Figs. 1–7.
Supplementary Data 1
Microrespirator Materials
Supplementary Data 2
Smoke Generator Materials
Supplementary Data 3
Supplementary Design Files: SmokingRobot.zip
Supplementary Video 1
Biomimetic Smoking Robot For In Vitro Inhalation Exposure Compatible With Microfluidic Organ Chips
Rights and permissions
About this article
Cite this article
Benam, K.H., Novak, R., Ferrante, T.C. et al. Biomimetic smoking robot for in vitro inhalation exposure compatible with microfluidic organ chips. Nat Protoc 15, 183–206 (2020). https://doi.org/10.1038/s41596-019-0230-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-019-0230-y
This article is cited by
-
Clinical Biomarkers to Evaluate Next Generation Inhaled Tobacco Products’ Effect in the Oral Cavity: a Narrative Review
Current Oral Health Reports (2024)
-
Recent advances in lung-on-a-chip technology for modeling respiratory disease
Bio-Design and Manufacturing (2023)
-
Human pituitary development and application of iPSCs for pituitary disease
Cellular and Molecular Life Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.