Abstract
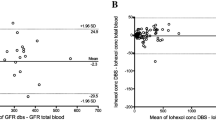
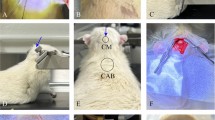
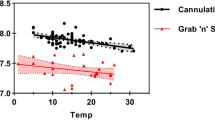
In many rodent laboratories, blood samples are collected from rats using the tail vein nick procedure and analyzed to quantify blood corticosterone levels as an indicator of stress. The standard method of corticosterone quantification often requires the collection of a relatively large volume of blood, followed by the extraction of the blood plasma. An alternative blood sampling method requires the collection of only a drop of blood on paper (the 'drop' method), minimizing handling of the animals, and does not require plasma extraction. The authors aimed to validate the drop method of blood sampling for use in corticosterone quantification. They induced stress in rats by cerebral ischemia, collected blood samples at various intervals using both the drop method and the plasma extraction method and then quantified corticosterone by radioimmunoassay. Corticosterone levels of the ischemic rats were compared with those of sham-operated rats and those of ischemic rats that had been given metyrapone, a glucocorticoid synthesis inhibitor, prior to vessel occlusion. Blood corticosterone levels in the samples obtained from the same animal using the two different methods were highly correlated for all rats. The authors further provide a regression model that can be used to predict plasma corticosterone values from those obtained from the drop blood samples. Quantification of corticosterone from only a small drop of blood has many practical and ethical advantages and should be considered as an alternative to standard methods.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sorrells, S.F., Caso, J.R., Munhoz, C.D. & Sapolsky, R.M. The stressed CNS: when glucocorticoids aggravate inflammation. Neuron 64, 33–39 (2009).
Rodrigues, S.M., LeDoux, J.E. & Sapolsky, R.M. The influence of stress hormones on fear circuitry. Annu. Rev. Neurosci. 32, 289–313 (2009).
Lee, A.L., Ogle, W.O. & Sapolsky, R.M. Stress and depression: possible links to neuron death in the hippocampus. Bipolar Disord. 4, 117–128 (2002).
McEwen, B.S. & Sapolsky, R.M. Stress and cognitive function. Curr. Opin. Neurobiol. 5, 205–216 (1995).
Sapolsky, R.M. Stress and plasticity in the limbic system. Neurochem. Res. 28, 1735–1742 (2003).
Khan, S., Milot, M., Lecompte-Collin, J. & Plamondon, H. Time-dependent changes in CRH concentrations and release in discrete brain regions following global ischemia: effects of MK-801 pretreatment. Brain Res. 1016, 48–57 (2004).
Hwang, I.K. et al. Ischemia-related changes of adrenocorticotropic hormone immunoreactivity and its protective effect in the gerbil hippocampus after transient forebrain ischemia. Neuroscience 126, 871–877 (2004).
Pulsinelli, W.A. & Brierley, J.B. A new model of bilateral hemispheric ischemia in the unanesthetized rat. Stroke 10, 267–272 (1979).
Pulsinelli, W.A., Brierley, J.B. & Plum, F. Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann. Neurol. 11, 491–498 (1982).
Sapolsky, R.M. & Pulsinelli, W.A. Glucocorticoids potentiate ischemic injury to neurons: therapeutic implications. Science 229, 1397–1400 (1985).
Milot, M.R. & Plamondon, H. Changes in HPA reactivity and noradrenergic functions regulate spatial memory impairments at delayed time intervals following cerebral ischemia. Horm. Behav. 59, 594–604 (2011).
Risedal, A., Nordborg, C. & Johansson, B.B. Infarct volume and functional outcome after pre- and postoperative administration of metyrapone, a steroid synthesis inhibitor, in focal brain ischemia in the rat. Eur. J. Neurol. 6, 481–486 (1999).
Wood, E.R., Mumby, D.G., Pinel, J.P. & Phillips, A.G. Impaired object recognition memory in rats following ischemia-induced damage to the hippocampus. Behav. Neurosci. 107, 51–62 (1993).
Block, F. & Schwarz, M. Global ischemic neuronal damage relates to behavioural deficits: a pharmacological approach. Neuroscience 82, 791–803 (1998).
Milot, M.R. & Plamondon, H. Time-dependent effects of global cerebral ischemia on anxiety, locomotion, and habituation in rats. Behav. Brain. Res. 200, 173–180 (2009).
Fluttert, M., Dalm, S. & Oitzl, M.S. A refined method for sequential blood sampling by tail incision in rats. Lab. Anim. 34, 372–378 (2000).
Vahl, T.P. et al. Comparative analysis of ACTH and corticosterone sampling methods in rats. Am. J. Physiol. Endocrinol. Metab. 289, E823–E828 (2005).
Smith-Swintosky, V.L. et al. Metyrapone, an inhibitor of glucocorticoid production, reduces brain injury induced by focal and global ischemia and seizures. J. Cereb. Blood Flow Metab. 16, 585–598 (1996).
Krugers, H.J., Maslam, S., Korf, J., Joels, M. & Holsboer, F. The corticosterone synthesis inhibitor metyrapone prevents hypoxia/ischemia-induced loss of synaptic function in the rat hippocampus. Stroke 31, 1162–1172 (2000).
Abatan, O.I., Welch, K.B. & Nemzek, J.A. Evaluation of saphenous venipuncture and modified tail-clip blood collection in mice. J. Am. Assoc. Lab Anim. Sci. 47, 8–15 (2008).
Brown, C. Blood collection from the tail of a rat. Lab Anim. (NY) 35, 24–25 (2006).
Acknowledgements
We sincerely thank Sylvie Émond for excellent technical assistance. This work was supported by a grant from the Natural Sciences and Engineering Research Council of Canada to H.P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Milot, M., James, J., Merali, Z. et al. A refined blood collection method for quantifying corticosterone. Lab Anim 41, 77–83 (2012). https://doi.org/10.1038/laban0312-77
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/laban0312-77